Post by icemandios on Mar 1, 2023 14:25:11 GMT
February 28, 2023 01:55 PM ESTUpdated 12 hours ago R&DOutsourcingPharma
Pfizer was warned of Lyme study problems months ago, trial research startup's CEO tells employees
Drew Armstrong
Executive Editor
Kyle LaHucik
Associate Editor
A contract research startup working on Pfizer’s Lyme vaccine trial alerted the drugmaker to problems with the study last year, but was told to keep the work moving to avoid raising public concerns about vaccine safety, the startup’s CEO has privately told employees in recent days.
Up until last week, Reify Health’s trials business, Care Access, was running more than half the US sites for Pfizer’s final-stage Lyme trial, and had enrolled thousands of people. In the days since Pfizer yanked the richly valued startup off the work, Reify CEO Ralph Passarella has told employees that his company months ago asked to pause its portion of the trial while it investigated issues.

Ralph Passarella
According to internal remarks to employees reviewed by Endpoints News, Passarella said his company late last summer told Pfizer that “we think we need to pause all operations while we think through” problems it had found. The remarks reviewed by Endpoints don’t say what those problems were, though Pfizer and Care Access both say they didn’t involve vaccine safety.
But according to Passarella, Pfizer told the startup that it couldn’t pause its portion “because if this gets out to the press then it’ll sound like there’s a safety issue with the vaccine.” That was in part because Care Access made up such a large portion of the trial, he said.
In response to questions from Endpoints, Pfizer said that it began a review of the trial as soon as it was told by Care Access about potential violations of good clinical practice, or GCP — shorthand for quality or ethical issues. It also said that it “conducted a thorough and methodical quality review of the operations and data collection practices.”
“During this review, additional potential violations of GCP at the trial sites operated by this third party were discovered, prompting further review,” Pfizer spokesman Kit Longley said in an email. The company didn’t comment on whether it was worried about public perception of a trial halt.
Pfizer also wouldn’t say when it was notified by Care Access, or what the issues were. But on Feb. 17, six months after Passarella said his company originally asked to pause its work, Pfizer announced that it was pulling thousands of participants from the Lyme trial – about half of the 7,000 people enrolled at that point, and all of the people Care Access had enrolled. Endpoints reported on Feb. 24 that Pfizer was removing Care Access from the trial entirely.
Reify declined to make Passarella available for an interview. In an emailed statement, Care Access CEO Ahmad Namvargolian declined to comment on the company’s conversations with Pfizer, calling them confidential and saying that Passarella’s remarks were taken out of context.
“Until its recent decision, Pfizer repeatedly and rightfully showed confidence in our ability to carry out the clinical trial,” Namvargolian said. “We continue to stand by the work we did on the trial.” Care Access has said that it has shared its side of the story with the FDA.
But there are indications that the startup was still having problems months later at a trial site in New Hampshire that experienced “an emergency,” according to an account from one participant. (Care Access says that other issues were not widespread.) And Passarella’s comments raise more questions about exactly what happened between a richly funded startup that promised to change how clinical trials are run, and one of the biggest drugmakers in the world.
Big promises, rich valuation
Reify Health was launched in 2012, and since its founding has raised $470 million. Its most recent round pulled in $220 million at a $4.8 billion valuation, with the promise that it could work with drugmakers to run clinical trials faster and more equitably than the contract research organizations, or CROs, that dominate the field.
Reify has two divisions, a tech arm known as OneStudyTeam, and its contract research unit, Care Access, which launched in 2016. When announcing the most recent round of funding last year, Namvargolian said that the company was “building the next generation of clinical trial infrastructure — the roads and highways of where clinical trials can go and who they reach.”
It works with some of the biggest names in the drug industry. Along with Pfizer, records show that it runs trials for drugmakers including Eli Lilly, Johnson & Johnson’s Janssen unit, Moderna and Amgen. Those companies either declined to comment or have said they’re monitoring the situation.
Pfizer’s Lyme disease trial represented a meaningful chance to deliver on the startup’s promise. Phase III vaccine trials often need thousands of patients, but the areas where Lyme disease is most prevalent — the backwoods of the Northeast — were far from the large, sophisticated medical centers where trials are more commonly run. Care Access promised to get Pfizer’s shot into smaller clinics and communities that had been underserved by trials, and to help Pfizer quickly enroll the study, with the goal of getting the shot to the FDA for approval by 2025.
Failure to push back
The vaccine test planned to enroll as many as 18,000 people, according to records on ClinicalTrials.gov, a US government website that tracks clinical trials. A delay would have created problems — there is a limited winter window to enroll patients, in between tick seasons.
Passarella said the company should have pushed back harder on Pfizer and to pause the work. In the remarks reviewed by Endpoints, he called that a mistake.
If Pfizer worried about the public perception of a trial pause, it was with some reason. In 1998, the FDA approved a vaccine for Lyme disease called LYMErix, produced by what is now GSK. The trial data showed that it was effective — cutting disease by 76% in the year after vaccination, according to a review article published by the journal Epidemiology & Infection.
But media reports of side effects, followed by lawsuits, doomed the shot. Some patients began to complain they developed joint aches and arthritis after being vaccinated. The FDA reviewed the safety data from the trial, as well as longer-term follow-up reports, and found no association between vaccination and longer-term side effects. But it was enough to turn the vaccine into a commercial failure. By 2001, sales had plunged. The next year, the company pulled the shot from the market, according to the journal.
‘An emergency’
While Passarella said Care Access went to Pfizer in August about problems it was having, there are indications that Care Access is still running into issues months later as the trial progressed.
Daniel Himmelstein, of Hanover, NH, signed up for the Lyme vaccine study and was scheduled to get his first dose in mid-November almost 100 miles away in Londonderry, he said in an interview. He’s an avid outdoor enthusiast and works in drug development.
He said he “wanted to see how trials operate from the patient perspective since I’m kind of in the industry.” Endpoints confirmed Himmelstein’s participation in the study via a review of communications and documents he received from Care Access.
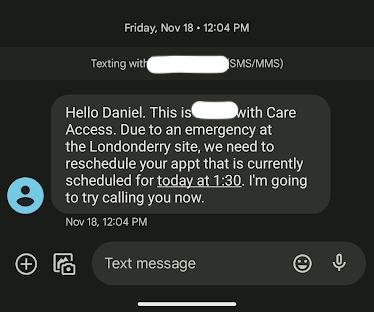
A Care Access representative (name redacted) informed Lyme disease vaccine trial participant Daniel Himmelstein of a study site “emergency” in November 2022, necessitating a rescheduling of his vaccine dose. Screenshot provided by Himmelstein
But as his appointment approached, he was told that it was going to be delayed by a week, according to a copy of a text message he provided to Endpoints: “Due to an emergency at the Londonderry site, we need to reschedule your appt that is currently scheduled for today.”
A week later, at the trial site, Himmelstein said staff told him there had been cases of carbon monoxide exposure because of poor ventilation of generators used to run the pop-up location.
“When the generators were set up, the nurse told me their exhaust was not either vented properly or somehow the carbon monoxide was getting back in,” Himmelstein said in an interview. “That’s the reason my appointment had been canceled.”
“There was no carbon monoxide poisoning at the Londonderry site, nor any other,” Care Access said in an emailed statement. Care Access said it confirmed that a generator exhaust had been placed too close to a tent, and that staff had smelled fumes.
“The principal investigator overseeing the site then suggested that staff members visit the hospital out of an abundance of caution,” the company said. It says it has now trained staff on how to avoid carbon monoxide poisoning risks in the future.
Pfizer didn’t provide a comment on the incident. Care Access declined to comment directly on Himmelstein’s participation.
Paperwork
That wasn’t the only issue, however. Himmelstein said he never received a scanned copy of his informed consent documentation that he requested. Separately, he received a letter about the trial that was addressed to another participant. And he said a reimbursement card for $50 he got didn’t activate because the date of birth differed from the one listed in the system used for trial management.
“There were some misaddressed notices that did not contain confidential information, but, as far as we know, no patient received confidential information about another patient,” Care Access said in its statement.
The company said it would be unfair to say that there were broad problems with its record-keeping: “We had rigorous policies and practices in place to ensure accurate record-keeping. Of course, isolated paperwork errors happen in any trial and, when they happened here, we always followed GCP standards in addressing them.”
As his second vaccination visit neared, Himmelstein said he was notified his appointment was canceled and would be rescheduled.
It never was, and he never received his second shot of the experimental vaccine. Instead, after Pfizer ended the Care Access portion of the work, Himmelstein got an email from the drugmaker saying that there had been problems with the company running his part of the trial: “Unfortunately, your study site is one of the sites operated by this third party, and this means that your participation in the study will need to end early.”
Pfizer was warned of Lyme study problems months ago, trial research startup's CEO tells employees
Drew Armstrong
Executive Editor
Kyle LaHucik
Associate Editor
A contract research startup working on Pfizer’s Lyme vaccine trial alerted the drugmaker to problems with the study last year, but was told to keep the work moving to avoid raising public concerns about vaccine safety, the startup’s CEO has privately told employees in recent days.
Up until last week, Reify Health’s trials business, Care Access, was running more than half the US sites for Pfizer’s final-stage Lyme trial, and had enrolled thousands of people. In the days since Pfizer yanked the richly valued startup off the work, Reify CEO Ralph Passarella has told employees that his company months ago asked to pause its portion of the trial while it investigated issues.

Ralph Passarella
According to internal remarks to employees reviewed by Endpoints News, Passarella said his company late last summer told Pfizer that “we think we need to pause all operations while we think through” problems it had found. The remarks reviewed by Endpoints don’t say what those problems were, though Pfizer and Care Access both say they didn’t involve vaccine safety.
But according to Passarella, Pfizer told the startup that it couldn’t pause its portion “because if this gets out to the press then it’ll sound like there’s a safety issue with the vaccine.” That was in part because Care Access made up such a large portion of the trial, he said.
In response to questions from Endpoints, Pfizer said that it began a review of the trial as soon as it was told by Care Access about potential violations of good clinical practice, or GCP — shorthand for quality or ethical issues. It also said that it “conducted a thorough and methodical quality review of the operations and data collection practices.”
“During this review, additional potential violations of GCP at the trial sites operated by this third party were discovered, prompting further review,” Pfizer spokesman Kit Longley said in an email. The company didn’t comment on whether it was worried about public perception of a trial halt.
Pfizer also wouldn’t say when it was notified by Care Access, or what the issues were. But on Feb. 17, six months after Passarella said his company originally asked to pause its work, Pfizer announced that it was pulling thousands of participants from the Lyme trial – about half of the 7,000 people enrolled at that point, and all of the people Care Access had enrolled. Endpoints reported on Feb. 24 that Pfizer was removing Care Access from the trial entirely.
Reify declined to make Passarella available for an interview. In an emailed statement, Care Access CEO Ahmad Namvargolian declined to comment on the company’s conversations with Pfizer, calling them confidential and saying that Passarella’s remarks were taken out of context.
“Until its recent decision, Pfizer repeatedly and rightfully showed confidence in our ability to carry out the clinical trial,” Namvargolian said. “We continue to stand by the work we did on the trial.” Care Access has said that it has shared its side of the story with the FDA.
But there are indications that the startup was still having problems months later at a trial site in New Hampshire that experienced “an emergency,” according to an account from one participant. (Care Access says that other issues were not widespread.) And Passarella’s comments raise more questions about exactly what happened between a richly funded startup that promised to change how clinical trials are run, and one of the biggest drugmakers in the world.
Big promises, rich valuation
Reify Health was launched in 2012, and since its founding has raised $470 million. Its most recent round pulled in $220 million at a $4.8 billion valuation, with the promise that it could work with drugmakers to run clinical trials faster and more equitably than the contract research organizations, or CROs, that dominate the field.
Reify has two divisions, a tech arm known as OneStudyTeam, and its contract research unit, Care Access, which launched in 2016. When announcing the most recent round of funding last year, Namvargolian said that the company was “building the next generation of clinical trial infrastructure — the roads and highways of where clinical trials can go and who they reach.”
It works with some of the biggest names in the drug industry. Along with Pfizer, records show that it runs trials for drugmakers including Eli Lilly, Johnson & Johnson’s Janssen unit, Moderna and Amgen. Those companies either declined to comment or have said they’re monitoring the situation.
Pfizer’s Lyme disease trial represented a meaningful chance to deliver on the startup’s promise. Phase III vaccine trials often need thousands of patients, but the areas where Lyme disease is most prevalent — the backwoods of the Northeast — were far from the large, sophisticated medical centers where trials are more commonly run. Care Access promised to get Pfizer’s shot into smaller clinics and communities that had been underserved by trials, and to help Pfizer quickly enroll the study, with the goal of getting the shot to the FDA for approval by 2025.
Failure to push back
The vaccine test planned to enroll as many as 18,000 people, according to records on ClinicalTrials.gov, a US government website that tracks clinical trials. A delay would have created problems — there is a limited winter window to enroll patients, in between tick seasons.
Passarella said the company should have pushed back harder on Pfizer and to pause the work. In the remarks reviewed by Endpoints, he called that a mistake.
If Pfizer worried about the public perception of a trial pause, it was with some reason. In 1998, the FDA approved a vaccine for Lyme disease called LYMErix, produced by what is now GSK. The trial data showed that it was effective — cutting disease by 76% in the year after vaccination, according to a review article published by the journal Epidemiology & Infection.
But media reports of side effects, followed by lawsuits, doomed the shot. Some patients began to complain they developed joint aches and arthritis after being vaccinated. The FDA reviewed the safety data from the trial, as well as longer-term follow-up reports, and found no association between vaccination and longer-term side effects. But it was enough to turn the vaccine into a commercial failure. By 2001, sales had plunged. The next year, the company pulled the shot from the market, according to the journal.
‘An emergency’
While Passarella said Care Access went to Pfizer in August about problems it was having, there are indications that Care Access is still running into issues months later as the trial progressed.
Daniel Himmelstein, of Hanover, NH, signed up for the Lyme vaccine study and was scheduled to get his first dose in mid-November almost 100 miles away in Londonderry, he said in an interview. He’s an avid outdoor enthusiast and works in drug development.
He said he “wanted to see how trials operate from the patient perspective since I’m kind of in the industry.” Endpoints confirmed Himmelstein’s participation in the study via a review of communications and documents he received from Care Access.

A Care Access representative (name redacted) informed Lyme disease vaccine trial participant Daniel Himmelstein of a study site “emergency” in November 2022, necessitating a rescheduling of his vaccine dose. Screenshot provided by Himmelstein
But as his appointment approached, he was told that it was going to be delayed by a week, according to a copy of a text message he provided to Endpoints: “Due to an emergency at the Londonderry site, we need to reschedule your appt that is currently scheduled for today.”
A week later, at the trial site, Himmelstein said staff told him there had been cases of carbon monoxide exposure because of poor ventilation of generators used to run the pop-up location.
“When the generators were set up, the nurse told me their exhaust was not either vented properly or somehow the carbon monoxide was getting back in,” Himmelstein said in an interview. “That’s the reason my appointment had been canceled.”
“There was no carbon monoxide poisoning at the Londonderry site, nor any other,” Care Access said in an emailed statement. Care Access said it confirmed that a generator exhaust had been placed too close to a tent, and that staff had smelled fumes.
“The principal investigator overseeing the site then suggested that staff members visit the hospital out of an abundance of caution,” the company said. It says it has now trained staff on how to avoid carbon monoxide poisoning risks in the future.
Pfizer didn’t provide a comment on the incident. Care Access declined to comment directly on Himmelstein’s participation.
Paperwork
That wasn’t the only issue, however. Himmelstein said he never received a scanned copy of his informed consent documentation that he requested. Separately, he received a letter about the trial that was addressed to another participant. And he said a reimbursement card for $50 he got didn’t activate because the date of birth differed from the one listed in the system used for trial management.
“There were some misaddressed notices that did not contain confidential information, but, as far as we know, no patient received confidential information about another patient,” Care Access said in its statement.
The company said it would be unfair to say that there were broad problems with its record-keeping: “We had rigorous policies and practices in place to ensure accurate record-keeping. Of course, isolated paperwork errors happen in any trial and, when they happened here, we always followed GCP standards in addressing them.”
As his second vaccination visit neared, Himmelstein said he was notified his appointment was canceled and would be rescheduled.
It never was, and he never received his second shot of the experimental vaccine. Instead, after Pfizer ended the Care Access portion of the work, Himmelstein got an email from the drugmaker saying that there had been problems with the company running his part of the trial: “Unfortunately, your study site is one of the sites operated by this third party, and this means that your participation in the study will need to end early.”





